Įvgeny Fesenko, Stanislav Kolesnikov, and Arkady Lyubarsky discovered in 1985 that cyclic guanosine monophosphate (cGMP) can initiate the photoresponse in rods. Further, it was proved that this factor reverted to 5’-AMP when it was inactivated. The “active factor” that the hormones produced was finally purified in 1958, and then identified as containing a ribose, a phosphate, and an adenine in equal ratios. While investigating the action of adrenaline on glycogenolysis the next year, Sutherland and Walter Wosilait reported that inorganic phosphate is released when the enzyme liver phosphorylase is inactivated but when it is activated, it incorporates a phosphate. In 1956 Edwin Krebs and Edmond Fischer discovered that adenosine triphosphate (ATP) is required for the conversion of glycogen phosphorylase b to glycogen phosphorylase a. Earl Sutherland investigated the effect of the hormones adrenaline and glucagon on glycogen phosphorylase, earning him the Nobel Prize in Physiology or Medicine in 1971. Glycogen phosphorylase catalyzes the first step in glycogenolysis, the process of breaking glycogen into its substituent glucose parts. A number of incremental but important discoveries through the 1950s added to their research, primarily focusing on the activity of glycogen phosphorylase in dog liver. The understanding of the concept of second messengers, and in particular the role of cyclic nucleotides and their ability to relay physiological signals to a cell, has its origins in the research of glycogen metabolism by Carl and Gerty Cori, for which they were awarded a Nobel Prize in Physiology or Medicine in 1947. Although more than 50 years have passed since their initial discovery, interest in cyclic nucleotides and their biochemical and physiological significance continues. The role of cyclic uridine monophosphate (cUMP) is even less well known.ĭiscovery of cyclic nucleotides has contributed greatly to the understanding of kinase and phosphatase mechanisms, as well as protein regulation in general. cAMP and cGMP are currently the most well documented cyclic nucleotides, however there is evidence that cCMP (with cytosine) is also involved in eukaryotic cellular messaging. They have been identified as secondary messengers in both hormone and ion-channel signalling in eukaryotic cells, as well as allosteric effector compounds of DNA binding proteins in prokaryotic cells. Their biological significance includes a broad range of protein- ligand interactions. As can be seen in the cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) images, the 'cyclic' portion consists of two bonds between the phosphate group and the 3' and 5' hydroxyl groups of the sugar, very often a ribose. Like other nucleotides, cyclic nucleotides are composed of three functional groups: a sugar, a nitrogenous base, and a single phosphate group. The cyclic portion refers to the two single bonds between the phosphate group and the riboseĪ cyclic nucleotide (cNMP) is a single- phosphate nucleotide with a cyclic bond arrangement between the sugar and phosphate groups. 1 It also serves as an extracellular morphogen for some slime moulds.Cyclic nucleic acid Cyclic adenosine monophosphate. In bacteria, cAMP is involved in the positive regulation of bacterial lac operon in association with the culture medium used for bacterial growth. Its activation of calcium channels gives rise to a minor pathway through which growth hormone-releasing hormone facilitates the release of growth hormones. 1Īpart from the intracellular signal transduction with certain kinases, cAMP has also a role in the regulation of the passage of calcium ions through ion channels. 
For instance, protein kinase C is not cAMP-dependent. Not all protein kinases though respond to cAMP. phosphorylate) specific proteins bound to the promoter regions of DNA, and thereby further enhance transcription. Upon phosphorylation, the protein substrates may act on the ion channels or they may become inhibited. The catalytic sub-unit may act on certain protein substrates, transferring phosphate to the latters’ serine or threonine residues. 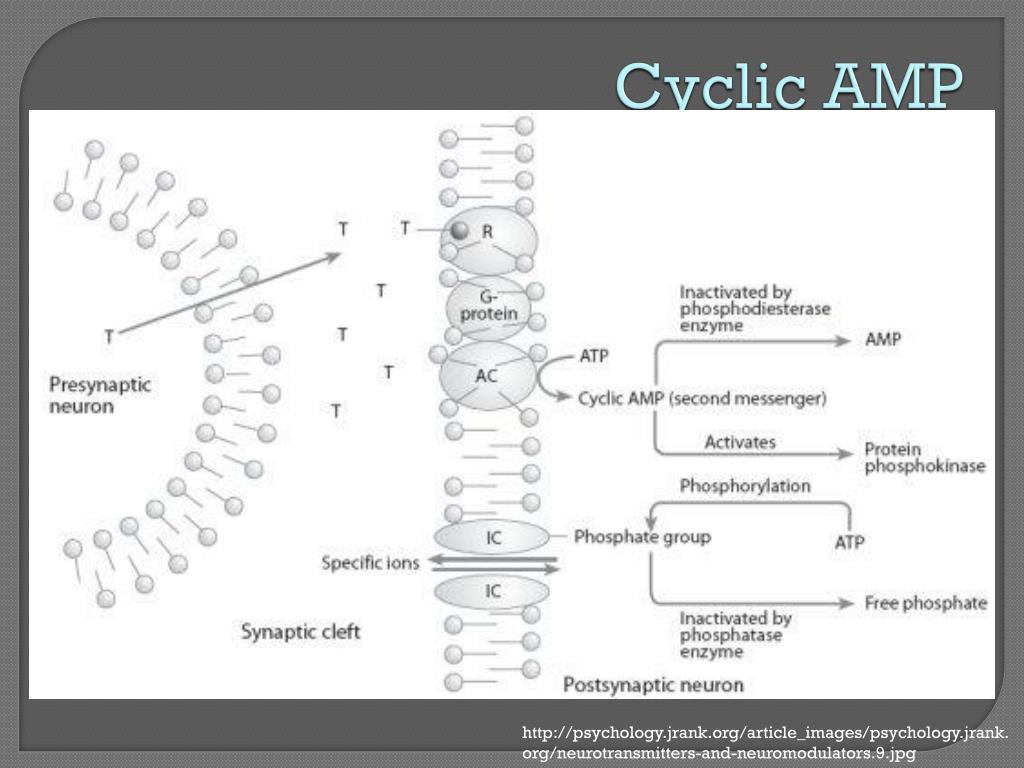
cAMP binds at the regulatory sub-units of the protein kinase, which leads to the dissociation of the latter into regulatory and catalytic sub-units. It works by activating protein kinase A (PKA). These hormones cannot pass through the plasma membrane of target cells and therefore need cAMP as a second messenger. In particular, it is used in transferring into cells the effects of glucagon and adrenaline. It is used for intracellular signal transduction in many different organisms, conveying the cAMP-dependent pathway. CAMP serves as a second messenger for signal transduction at the cellular level. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
